Development
Latest News
Latest Videos

More News

The new collaboration will aim to streamline formulation-to-manufacturing workflows and de-risk development.
FDA’s internal findings raise new concerns over pediatric vaccine safety and signal potential regulatory shifts affecting future COVID-19 vaccine development.

How is the biopharma industry solving manufacturing and scale-up bottlenecks for cell and gene therapies? Read on to find out.
The transformative impact of complex biopharmaceuticals hinges on integrating advanced delivery systems, often accelerated by AI modeling, with robust manufacturing practices and regulatory alignment to ensure stability, safety, and effective patient access.
Samsung Life Science Fund’s investment in Phrontline Biopharma highlights growing industry momentum behind dual-payload ADCs.

Progress in vaccine development is closely tied to advancements in biopharmaceutical technologies, regulatory landscapes, and quality assurance methodologies.

Regulators and manufacturers face new uncertainty as shifting CDC vaccine language raises concerns for evidence-based oversight and biopharma compliance.

Sapient said the workflow will allow for direct measurement of more than 10,000 protein groups in a single, 5-µm slide.

RxCelerate and Constructive Bio will combine programmable biology with integrated discovery tools to speed engineered biologics for hard-to-treat diseases.

Webinar Date/Time: Tue, Dec 9, 2025 11:00 AM EST

The new strategic collaboration seeks to develop up to four first-in-class myeloid cell engagers offering broad and safe treatment for blood and solid cancers.
The Switch to Cell-Free DNA: Strategic and Regulatory Considerations for Advanced Therapies
Webinar Date/Time: Thu, Dec 11, 2025 11:00 AM EST

Biogen reports CHMP support for an enhanced nusinersen regimen, signaling potential shifts in SMA treatment strategies and global regulatory decisions.

Experts at AAPS PharmSci 360 discuss industry-wide transformation from AI implementation and the ripple effect on workforce development.

Government and academic leaders say that to prevent catastrophic loss of expertise and investment, coordinated policy and significant investment in workforce talent are urgently needed in the UK.

Help shape industry insights. Take our survey on the biopharma impacts of the FDA Commissioner’s National Priority Vouchers program.

Webinar Date/Time: Tue, Dec 9, 2025 2:00 PM EST

FDA’s newly introduced review route enables faster clearance of custom genetic and cell treatments, reshaping rare-disease development and manufacturing strategies.

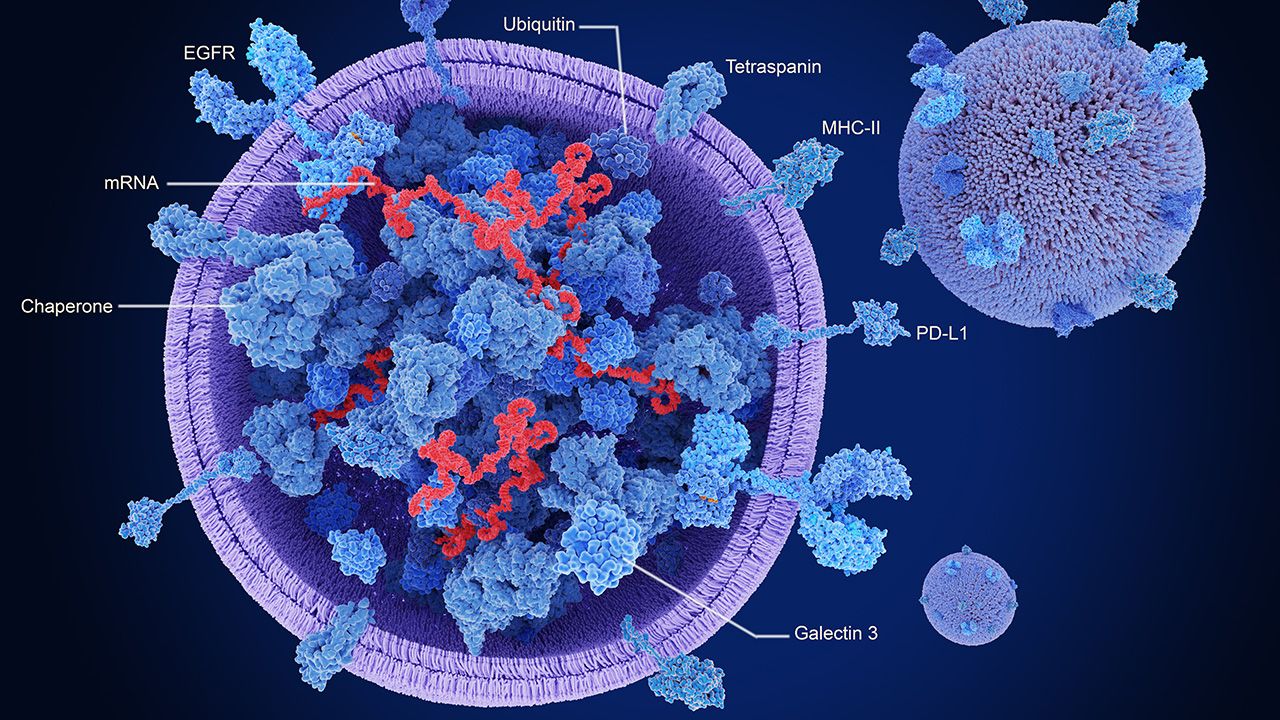
The company demonstrates advances in glioma hydrogel, high-concentration biologics, and nanoencorafenib licensing, impacting drug delivery strategies.

At AAPS PharmSci 360, Kelly Donovan says tailored analytical strategies are necessary for reducing development delay risks.

Webinar Date/Time: Thu, Nov 20, 2025 12:30 PM EST

Financing strategies for new modalities and AI will be central at PharmSci 360, shaping how advanced therapeutics move toward global patient access.
A new Cue Biopharma–ImmunoScape partnership seeks to advance targeted TCR-T expansion for solid tumors, supporting broader access and improved clinical durability.

The second installment of the monthly BioPharma By the Numbers infographic series details the hiring and retention challenges faced by US biopharma.
Codexis and Nitto Denko Avecia aim to advance next-gen siRNA production methods for improving scalability of RNA therapeutics.