
Manufacturing Equipment, Biologics
Latest News

Latest Videos

More News

This paper outlines a systematic approach to detecting contamination through process deviations, including changes in % dissolved oxygen, pH, and metabolic patterns.

Nicole Hunter, head of Global WMArchitect at WMFTS, sat with the PharmTech Group at INTERPHEX 2025 to discuss the industry initiative around single-use assembly components that was prompted by the supply disruptions caused by the COVID-19 pandemic.

BioPure’s BioClamp connector has been upgraded and is now manufactured to be 13% lighter than the previous model, allowing users to achieve a 26% reduction in carbon dioxide emissions across the full lifecycle of a product.

The addition of the two larger-volume bioreactor sizes to Cytiva’s Xcellerex X-platform portfolio will allow scientists and researchers to scale up from 50 L to 2000 L to meet growing demand for clinical trials and regulatory approved products.

Advances in automation technologies and bioprocessing equipment are driving fuller adoption of automation in biomanufacturing.

Operating in an aging facility and/or utilizing outdated equipment may lead to drug shortages and high operating costs.

With financing led by OrbiMed, Novo Holdings, and Jeito Capital, Alentis Therapeutics will develop a pipeline of Claudin-1-targeted ADCs to treat solid tumors.
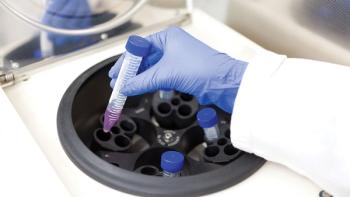
This case study describes how the risk assessment of the first-to-market single-use disc-stack centrifuge was conducted.

Freudenberg Medical now offers custom single-use assemblies to expand its offerings in silicone extrusion and molding for biopharma applications.

Under the expanded agreement, Univercells and University of Pennsylvania will evaluate certain bioreactors for scalable gene therapy production.

AGC Biologics has completed an expansion at its Copenhagen, Denmark, site, which doubles its bioreactor capacity for mammalian-based cell culture.

Under a partnership, ProPharm and PBL have launched the Cell Factory Box, an enclosed and fully automated cell factory manufacturing device.

ADC manufacturing facilities are requiring more stringent containment requirements as well as advanced analytical detection.

The Xcellerex magnetic mixer, single-use mixing system was designed to address challenges in large-scale mAb, vaccine, and genomic medicine manufacturing processes.

WMFTS’ new WMArchitect single-use solutions offers standard ready-to-use single-use assemblies as well as customized designs for biopharma fluid management.

Despite a growing trend toward single-use bioreactors, stainless-steel retains its relevance for certain products.

The evolution of therapeutic modalities drives the adoption of single-use technologies.

Challenges in fermentation can be addressed through equipment changes, facility design, and process development.

Purolite and Repligen have commercially launched a new CH1 affinity resin for the purification of specialized mAbs.

The authors give an overview of scaling approaches and present good scaling practices for the biopharma industry.

Pharmaceutical Technology Europe® spoke with Fiona Bellot, sales director, Cellexus BioreactorSystems, at the inaugural Start-Up Market on the show floor at CPHI Barcelona 2023.

As oligo manufacturers scale up production from lab-scale to clinical and commercial scales, they may require volumes of hazardous materials that drive additional facility design considerations.

SCHOTT now offers ViewCell, a new component meant to add flexibility to biopharmaceutical process control.

The site was acquired by AGC Biologics in July 2020, and the Milan location is the first cell and gene therapy site approved in Europe for GMP manufacturing of clinical and commercial supplies.

The bioreactors can integrate with Emerson’s DeltaV PK Controller in addition to S-Series and M-Series controllers.







