
Weak or faulty quality systems can hurt a company at every stage of a product’s lifecycle.

Weak or faulty quality systems can hurt a company at every stage of a product’s lifecycle.

The agency is offering patients with life-threatening diseases a how-to tool on accessing investigational drugs.

FDA sent a warning letter to Sage Products, Inc. regarding CGMP failures involving test methods, sampling, and sterilization procedures.

The agency will lead an international team to create a Supply Chain Security Toolkit of resources to educate the pharmaceutical and healthcare industries on supply chain vulnerabilities.

Updated ISPE guidance offers risk assessment guidance to avoid cross contamination in multiproduct pharmaceutical manufacturing facilities.

The agency recommended a total of 11 drugs at its July meeting, including five orphan drugs.

The agency cited the Italian company for aseptic processing failures.

The National Academies of Sciences, Engineering, and Medicine released a report on its findings on the opioid abuse epidemic, citing the need for a prescribing culture change.

BIO and PhRMA issued statements expressing the groups’ approval of the continuation of user fee programs.

The agency sent a warning letter to Vista Pharmaceuticals Limited for CGMP failures related to equipment cleaning and process control.

One day after Ocular Therapeutix sent FDA a detailed response highlighting manufacturing improvements, FDA rejected its application for Dextenza, a new drug designed to alleviate post-operative eye pain.

In an FDAVoice blog post, the CDER director highlighted two recent scientific advances.

The agency��’s Pharmacovigilance Risk Assessment Committee made recommendations on the risk of multiple sclerosis treatments, injectable methylprednisolone medicines, and the use of gadolinium contrast agents.

FDA sent a warning letter to Shandong Analysis and Test Center for CGMP violations including data integrity issues and insufficient testing procedures.
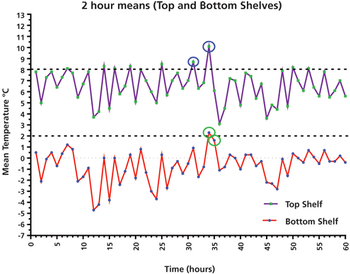
This column presents a data case study of a laboratory refrigerator and its qualification performance over five days, with important lessons for using average and individual results, as well as user requirements.

The opening presentation gives the company a chance to put their best foot forward, according to Siegfried Schmitt, principal at PAREXEL.

This article presents a general strategy for authorship of deviation investigations, with primary focus on regulatory inspection success.
While the measurement of the toxicity of leachables is not always a required parameter, the information collected during these studies could inform future bioprocessing runs.

Single-use bags containing toxic or hazardous materials required special handling.

CDER’s Janet Woodcock endorses modern drug manufacturing to ensure access to safe and reliable medicines.

Continuous processing of 100 g of monoclonal antibody in 24 hours has been demonstrated using lab-scale equipment.

Electronic systems can remove opportunities for individuals to make mistakes or to manipulate the data.

Optimize practices and meet requirements using electronic data integrity systems.
Short tandem repeat profiling is the most validated method to confirm cell line identity and avoid misidentified or cross-contaminated cell lines.

The agency announced a plan to eliminate its existing orphan designation request backlog.