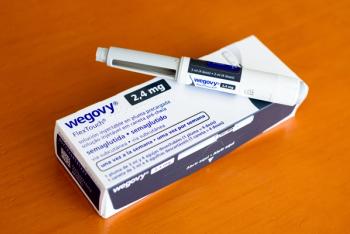
The European Commission has authorized a higher 7.2 mg once-weekly maintenance dose of semaglutide for adults with obesity, supported by Phase III STEP UP data showing greater weight loss versus placebo while preserving muscle function.

The European Commission has authorized a higher 7.2 mg once-weekly maintenance dose of semaglutide for adults with obesity, supported by Phase III STEP UP data showing greater weight loss versus placebo while preserving muscle function.

The approval of Uplizna (inebilizumab) for adults with anti-AChR or anti-MuSK antibody–positive generalized myasthenia gravis is supported by Phase III MINT data demonstrating significant improvements in MG-ADL and QMG scores, with twice-yearly maintenance dosing following initial loading infusions.

Where biotech partnerships shape the future of life sciences.

This event offers knowledge sharing, relationship building, and deal-making for biotech and pharmaceutical companies to advance commercialization of advanced therapies.

This event brings together leading chemists, biologists, toxicologists, CMC experts, regulatory specialists, and technology providers to discuss advances in next-generation therapeutics.

This event will enable biologics and therapeutic R&D experts build partnerships, align specialisms, bridge unknowns and commercially convert novel cures into life-changing outcomes for patients.

This event fuses industry innovation with an expert-led conference, where the newest ideas are shared, technology is unveiled, and the power of science though commercialization comes to life.

Sander Van Gessel, MEng, Business Unit Director, DFE Pharma, discusses the important components of nitrosamine impurities from a manufacturing point of view as a part of his session, "Reducing Nitrosamines Without the Use of Scavengers: The Critical Role of Excipients - An Excipient Manufacturer's View."

Sander Van Gessel, MEng, Business Unit Director, DFE Pharma, discusses the recent findings of nitrosamine impurities in human drugs as a part of his session, "Reducing Nitrosamines Without the Use of Scavengers: The Critical Role of Excipients - An Excipient Manufacturer's View."

Humberto Vega, PhD, Executive Director, Bristol Myers Squibb, discusses the requirements needed to be successful in global cell therapy as a part of his session, "Successfully Navigating Global Cell Therapy Requirements."

Humberto Vega, PhD, Executive Director, Bristol Myers Squibb, discusses the journey from clinical to commercial cell therapies as a part of his session, "Successfully Navigating Global Cell Therapy Requirements."

Bassem Gayed, PhD, Senior Technical Director of CAR-T Process Engineering, Manufacturing Science & Technology at Bristol Myers Squibb, discusses the importance of CAR-T as a modality and other highlights from his part of the session, "Process Validation for CAR-T Products: A Continuous Journey."

Catalent provides fill/finish services across a range of product formats from clinical to commercial scale.

Bassem Gayed, PhD, Senior Technical Director of CAR-T Process Engineering, Manufacturing Science & Technology at Bristol Myers Squibb, discusses the connection of process validation and CAR-T processes.

Maik Jornitz, Principal Consultant, BioProcess Resources LLC, discusses how to upgrade facilities in an efficient manner and other highlights from his presentation.

Kelley Burridge, PhD, Product Quality Team Leader, OBP, OPQ, CDER, U.S. FDA, discusses the importance of continuous manufacturing for the future.

Kelley Burridge, PhD, Product Quality Team Leader, OBP, OPQ, CDER, U.S. FDA, discusses the highlights of her ICH Q13 session that she wants others to be aware of.

Maik Jornitz, Principal Consultant, BioProcess Resources LLC, discusses the definition of patient safety and how to implement new technologies into upgraded facilities.

Kelley Burridge, PhD, Product Quality Team Leader, OBP, OPQ, CDER, U.S. FDA, discusses how advanced manufacturing technology can improve things such as cost and efficiency.

Kelley Burridge, PhD, Product Quality Team Leader, OBP, OPQ, CDER, U.S. FDA, discusses the barriers to the development of continuous manufacturing processes after implementing ICH Q13.

Part of the session "Transforming Medicine: Unleashing the Potential of Cell and Gene Therapies," Sadelain discusses CAR-T cells for immunotherapy and the future of cell/gene therapies.

The new guidance provides detailed recommendations to drug developers with a target of helping to ensure that drug developers provide adequate information to assess potency at each stage of a product’s life cycle.

Lilly will cap insulin prices for patients with and without insurance at $35 per month.

AstraZeneca has entered into a global exclusive license agreement with KYM Biosciences for CMG901, which is being investigated as a gastric cancer treatment.

Moderna and Merck’s investigational personalized mRNA cancer vaccine has been granted Breakthrough Therapy Designation by FDA.

WHO is appealing for US $392 million to reach 12.9 million people with essential health assistance in 2023.

Orchard Therapeutics has announced an agreement which will enable reimbursed access to Libmeldy for all eligible MLD patients in Sweden.

Bio-Rad’s newly launched StarBright Blue and StarBright Yellow dyes are intended for use in various flow cytometry applications.

Eurofins Viracor ExPeCT CAR-T qPCR assay is a tool designed for monitoring and optimizating CAR-T therapies.

Moderna will leverage Life Edit’s suite of gene editing technologies to advance development of in vivo gene editing therapies.

November 4th 2020

January 5th 2022

August 1st 2021