
The agency will lead an international team to create a Supply Chain Security Toolkit of resources to educate the pharmaceutical and healthcare industries on supply chain vulnerabilities.

The agency will lead an international team to create a Supply Chain Security Toolkit of resources to educate the pharmaceutical and healthcare industries on supply chain vulnerabilities.

A Merck, Pfizer, and Corning collaboration resulted in development of Corning Valor Glass for improved drug storage and delivery and will create US jobs.

Updated ISPE guidance offers risk assessment guidance to avoid cross contamination in multiproduct pharmaceutical manufacturing facilities.

GSK plans to invest in several UK manufacturing sites and to close its Slough site in conjunction with the sale of the Horlicks brand manufactured in Slough.

The addition of 20,000 sq. ft. of manufacturing and office space increases Avecia’s Milford, Ma, oligonucleotide capacity to 3.0 mol.

GSK plans to deliver up to 40 million doses of its quadrivalent flu vaccine for the 2017–2018 season.

Higher-flow peristaltic pumps from Watson-Marlow Fluid Technology Group are designed for upstream and downstream bioprocessing with single-use fluid path assemblies.

FDA advisory committee has recommended for approval Novartis CAR-T cell therapy CTL019 for the treatment of relapsed and refractory B-cell acute lymphoblastic leukemia in pediatric and young adult patients.

The acquisition will add recombinant-based influenza vaccine to Sanofi Pasteur’s portfolio

ABEC increases the maximum capacity of its Custom Single Run Bioreactors to 4000 L, doubling the industry standard.

The National Institutes of Health says a new study showed that hydroxychloroquine reduced the transmission of Zika in pregnant mice.

A $5.5 billion acquisition brings Capsugel’s oral dosage delivery capabilities to Lonza’s portfolio.

IDBS described benefits and best practices for laboratory electronic data systems.

This article presents a general strategy for authorship of deviation investigations, with primary focus on regulatory inspection success.

The authors summarize the current regulatory expectations regarding the number of PPQ batches required and provide potential approaches that can be used to determine and justify the number of PPQ batches.
While the measurement of the toxicity of leachables is not always a required parameter, the information collected during these studies could inform future bioprocessing runs.

Single-use bags containing toxic or hazardous materials required special handling.

CDER’s Janet Woodcock endorses modern drug manufacturing to ensure access to safe and reliable medicines.
The authors present a shift toward more integrated purification processes.

Perfusion processes can attractive for biologics drug manufacturing; however, obstacles remain.

Continuous processing of 100 g of monoclonal antibody in 24 hours has been demonstrated using lab-scale equipment.

The company has broken ground on a R&D and process development facility in Missouri.

Quattroflow’s EZ-Set Pump Chamber Replacing System for its single-use pumps allows users to change chambers quickly by hand.

Two recently-signed agreements will transfer Takeda’s measles and acellular pertussis vaccine technologies to India-based multinational company Biological E. Limited to develop low-cost combination vaccines including diphtheria, tetanus and acellular pertussis (DTaP), and measles-rubella (MR) vaccines.
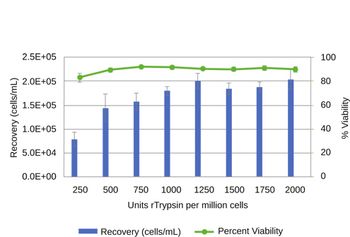
Traditional planar culture formats are being superceded by microcarriers for large-scale cell therapy manufacturing.