
Vetter held a groundbreaking ceremony for its new facility in Ravensburg.

Vetter held a groundbreaking ceremony for its new facility in Ravensburg.
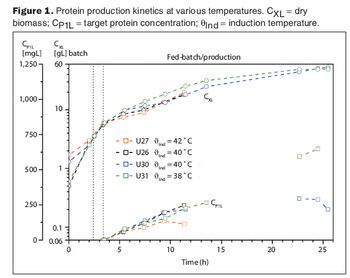
Two case studies show how advanced information technologies make process development more efficient.

International outsourcing and rising theft spur regulatory action and manufacturer oversight.

Broader benefits and biosimilars will offset hefty fees and discounts while preserving R&D incentives.
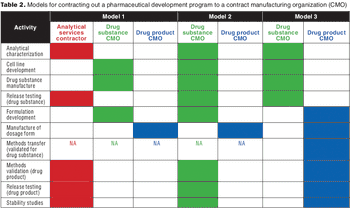
Formulation strategy is an important consideration when selecting and managing outsourced biopharmaceutical development programs.

Computational fluid dynamics can resolve performance problems.
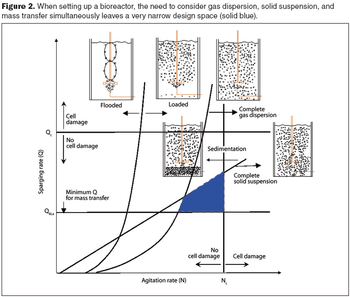
Computational fluid dynamics is a powerful tool to optimize processes.

The FDA is expanding postmarketing safety requirements, despite limited resources to manage these added responsibilities.

An effective CAPA plan provides a mechanism for responding to the unexpected.
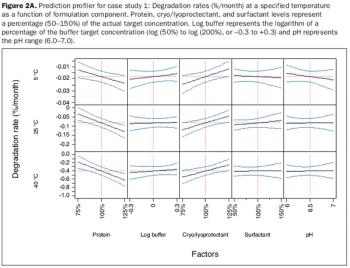
Design of experiments is a valuable tool for identifying aspects of a formulation that are critical to product quality.

The new Sentinel system aims to expand access to data on medical product safety and patient effects.
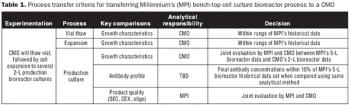
Sufficient process history is key to the rapid transfer of your process.

Trouble at Genzyme and with flu vaccine production illustrates the challenges in producing safe and potent biologics.

One might look at QbD's plodding growth and conclude that it is never going to make it to graduation.
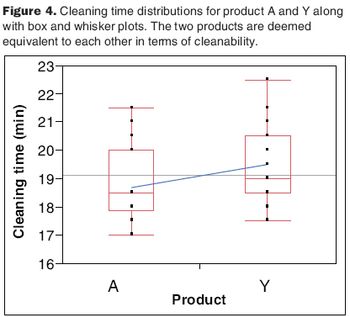
The two-one-sided t-test compares the equivalency of two data sets.

How will implementing Quality by Design strategies affect your compliance status?

Demand for new vaccines and therapies in 2010 will be offset by concerns about drug prices and product safety.

The nimbleness of biotechs makes them well suited to implementing QbD. Here's how to get started.

Regulatory flexibility can make continuous improvement possible.

A new analysis highlights growth opportunities and challenges for contract development and manufacturing organizations.
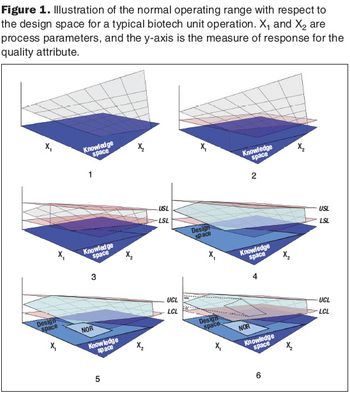
Second in a three-part series that discusses the complexities of QbD implementation in biotech development.

Vaccine research and development is surging, but continues to face manufacturing and regulatory challenges.
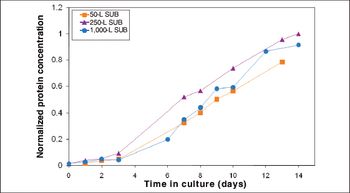
Process performance was comparable across all scales, and fiber optic sensors appeared interchangeable with conventional probes.

The focus on the design space will lead to a new workspace, and will affect staff in the development, manufacturing, and quality functions.

Heightened attention to product safety issues is slowing the approval process for new therapies.