
Every biotech company reaches a point in its development where it must decide what path it will take after it passes the start-up phase. This article discusses what the company must consider to decide what business model it will follow.
Senior Vice President of Operations at Tunnell Consulting

Every biotech company reaches a point in its development where it must decide what path it will take after it passes the start-up phase. This article discusses what the company must consider to decide what business model it will follow.
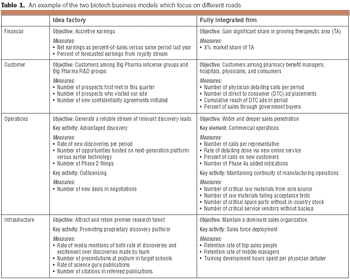
Two business models that companies can follow to map their strategy from concept to implementation.

Biotechs can avoid becoming a zombie by having a thorough business plan and a clear focus.

In the process of developing breakthrough biopharmaceuticals with profound therapeutic promise, the many detailed requirements for a successful investigational new drug (IND) submission may seem petty, but they are not. With an IND, you are essentially moving from the cloistered world of the laboratory into a highly regulated industry where details not only matter, but are also greatly magnified by the overriding requirements of safety and efficacy. Treat those details with forethought and you will eventually succeed. Treat them as an afterthought and all of your pioneering science, state-of-the-art technology, and therapeutic ambition could come to nothing. At the very least, your progress to market could be delayed significantly. And if, like most young biopharmaceutical companies, you are on a short financial leash, such delays can be fatal for securing additional funding.

Published: January 1st 2008 | Updated:

Published: January 1st 2008 | Updated:
Published: March 1st 2008 | Updated:

Published: September 1st 2008 | Updated: