
Pharmaceutical companies have shifted their approach to development of vaccines as a result of the COVID-19 pandemic.

Pharmaceutical companies have shifted their approach to development of vaccines as a result of the COVID-19 pandemic.

Key challenges posed to autologous and allogeneic treatments could be resolved by in-vivo CAR-T gene therapies.
Decreasing vein to vein time saves lives.

There is growing pressure for robust and economically scalable viral-vector manufacturing technologies.

Webinar Date/Time: Wed, Mar 8, 2023 11:00 AM EST

CMO growth is focused on analytical testing, toxicity testing, and fill/finish operations.

A slew of late-stage clinical trials is expected to push new regenerative medicines onto the market in the next few years.

Improvement in viral vector yield has become integral to new cell and gene therapy product development.
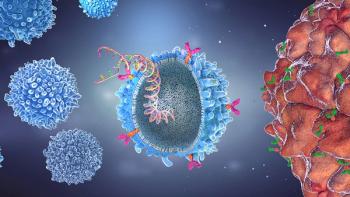
In vivo CAR-T gene therapies could overcome the challenges faced by autologous and allogeneic treatments.

This new pioneering phase of mRNA is loaded with potential, but also obstacles and false dawns.

The COVID-19 pandemic helped to showcase RNA molecules and their therapeutic potential. There were also lessons learned in terms of drug delivery, manufacturing, logistics, and storage.

Big data is a natural for pharmaceutical industry players that have not already embraced it.

Messenger RNA is inherently unstable and thus requires unique solutions to protect its cohesion.

This compilation allows readers to adjust their thinking to appreciate the full impact certain select technologies will make on the industry by 2026.

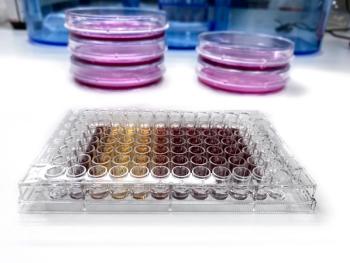
Biologic drug development requires relevant bioassays to measure and help predict cellular response.

Cell and gene therapies is unequivocally viewed as the biggest opportunity in the bio/pharma space.

Webinar Date/Time: Tue, Jan 24, 2023 11:00 AM EST

Sosei Heptares and Eli Lilly have entered a multi-target collaboration and license agreement in diabetes and metabolic diseases.

GSK and Wave Life Sciences have entered into a collaboration to drive drug discovery and development for novel genetic targets.

AbCellera and Rallybio are teaming up to discover, develop, and commercialize novel antibody-based therapeutics for rare diseases.

Emmes’ new facility is designed to support clients conducting cell and gene therapy research worldwide.

Particle engineering can be a resourceful tool to advance drug candidates by offering drug delivery innovation.

Deeper biological understanding and technology innovations have improved the fate of ADC development.

Through the integration of human expertise, artificial intelligence, and automation robotics, bio/pharma companies can more efficiently target drug discovery efforts with bigger payoff.

Generate Biomedicines published a preprint describing technology that can generate novel proteins towards desired functional or other properties.