
The new excipients manufacturing facility is expected to be completed in 2025.


The new excipients manufacturing facility is expected to be completed in 2025.

WuXi Biologics is expanding its manufacturing capacity for drug substance and drug product at its sites in Leverkusen and Wuppertal, Germany.

The market potential of nucleic acid-based therapies have been pushed into the spotlight following the success of the COVID-19 vaccines.

Guidance for GMP manufacture of emerging therapies has evolved to accommodate innovation and the broader spectrum of products in development, but greater harmonization is still needed.

Gregor Kawaletz, head of Oral Solid Dose Business Unit at Recipharm, discusses the immediate future of oral biologics

Avid Bioservices has launched new CGMP mammalian cell manufacturing suites at its Myford, Calif., facility.

The BalanCD CHO media portfolio is designed to ensure maximized growth, viability, and productivity of CHO cell lines.

The COVID-19 vaccines opened the door to powerful market potential for other nucleic acid-based therapies.

Samsung Biologics plans to invest more than ₩1.9 trillion (US$1.5 billion) in the new Incheon, South Korea, facility, which will hold a manufacturing capacity of 180,000 L.

Sandoz, a Novartis division, is expected to invest at least $400 million to build a new biologics manufacturing facility in Lendava, Slovenia, to support growing demand for biosimilars.

Under a technology licensing agreement with Bhami Research Laboratory, Catalent intends to develop formulations that will allow for the delivery of high-concentration biologics subcutaneously.
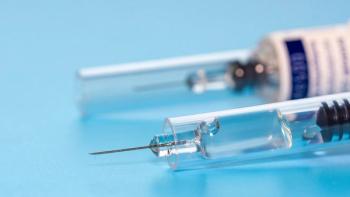
Shifts in pharmaceutical packaging have spurred tremendous growth in the pre-filled syringe fill/finish industry.
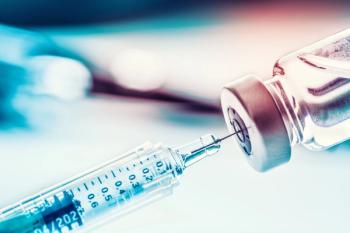
The trends shaping the growth of the biologics outsourcing industry demand attention.

There is an evolution in the way technology and best practices are blended to meet cold-chain supply requirements for temperature-sensitive biotherapeutics.

Alternative approaches to freeze drying are gaining popularity and will help to overcome some of the challenges associated with conventional lyophilization.

In this paper, the authors lay out some commonly accepted HCP analytical methods, the challenges of HCP assay, and provide recommendations on what can easily be accomplished in-house and when it may be better to outsource.

Efforts to optimize the ideal blend of technology and best practices that are needed to protect and deliver time- and temperature-sensitive biologic therapies, vaccines, and cell and gene therapies continue to evolve.

Thermo Fisher Scientific’s new facility in Hangzhou, China, is designed to boost biologics and sterile development and manufacturing capabilities in the Asia-Pacific region.

Digitalization of bioprocessing is slowly bringing benefits to the biopharma industry.
High-cost-per-dose large molecules can make a big impact on cell and gene therapy supply chain management.

Scorpion’s new facility is designed for the development and manufacturing of clinical and commercial-scale biologic drugs.

CBER maps modernization plan to handle surge in research and applications.

Experts divulge where the industry is outsourcing most frequently, when companies should outsource more, and when performing tasks in-house might be best.

WuXi STA is opening a new large-scale oligonucleotide and peptide manufacturing facility.

MilliporeSigma and Lotte Group will collaborate on facility design and workforce training for a new biologics business unit in the US.