
Automation, digitalization, and supply-chain strategies help mitigate vulnerabilities in both autologous and allogeneic cell therapy manufacturing.

Automation, digitalization, and supply-chain strategies help mitigate vulnerabilities in both autologous and allogeneic cell therapy manufacturing.

The company has made several moves in recent weeks to expand manufacturing capacity and cell line services.

Technologies in use and on the horizon could change aseptic processing in ways that seemed inconceivable years ago but approaches to aseptic process validation still need to move beyond their 1970s roots.

The new facility in Grand Rapids, MI, is part of the CDMO’s aggressive expansion plan.

Manufacturers and the US government are investing heavily in traditional and non-traditional forms and materials to ensure supplies of containers and delivery devices for COVID-19 treatments and vaccines.

Safeguarding against microbial contamination requires rapid detection and innovative technology.

The companies have entered into a manufacturing agreement for the fill finish supply of lenzilumab for the potential treatment of COVID-19.

Risk aversion is impeding the use of closed aseptic systems, and adding unnecessary cost and complexity to aseptic operations in the pharma industry.

Questions and answers about contemporary closed-system design criteria for aseptic processing equipment

The industry’s aversion to risk has led to its treating closed aseptic processing systems as miniature cleanrooms, resulting in redundant and expensive practices. This article ponders this impasse, examining new technologies and applications in light of past regulatory guidance and more than 40 years of operating evidence.

Catalent completes purchase of biologics fill-finish and oral solid dose facility in Anagni, Italy

US-based SP Industries acquired the sterile filling line assets of Spain-based i-Dositecno.

Gerresheimer enlarged a cleanroom, installed a high-performance furnace, and automated testing and packaging systems for pharmaceutical glass packaging in Essen, Germany.

Ireland's National Institute of Bioprocessing Research and Training (NIBRT), together with technology providers such as GE Healthcare, and universities in Ireland and around the world, are developing courses that aim to close the gap between theoretical studies and practical workplace needs.

Advances in fill/finish for parenteral packaging address demands for efficiency and product safety.

Nephron Pharmaceuticals is partnering with Clemson University to create a robotic solution for syringe-filling automation to enhance sterile manufacturing.

Lyophilization Services of New England acquired a sterile injectables manufacturing facility in León, Spain.

A US District Judge in the Southern District of Florida held that the US Stem Cell Clinic of Weston, FL and US Stem Cell of Sunrise, FL adulterated and misbranded a stem cell drug product made from a patient’s adipose tissue.
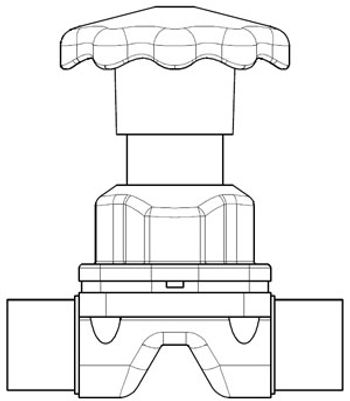
Valve design and materials affect performance and cost to maintain.

Sterile filtration is often required for biologics but presents degradation and compatibility challenges.

At INTERPHEX 2019, CEO Richard Johnson highlighted the importance of PDA’s new Asian business unit and outlined the organization’s plans. Data integrity guidance for manufacturing and quality systems will be published by the end of the year, as efforts move into big data and artificial intelligence.

Pharmaceutical Technology and BioPharm International will present a Keynote Session on Meeting Bioprocessing Manufacturing Capacity Demands on Wednesday, April 3, 2019, during INTERPHEX 2019 at the Javits Center in New York City.

In a keynote session at INTERPHEX 2019, experts will review and debate the issues and present potential solutions for contamination issues in aseptic manufacturing.

Distek, a provider of laboratory testing instruments, added a dual impeller single-use bioreactor (SUB) system to its BIOne portfolio.

The Pharma Services business of Thermo Fisher Scientific will invest $150 Million at three facilities.