
The companies are collaborating on the commercialization of two biosimilar candidates in the US and Canada.

The companies are collaborating on the commercialization of two biosimilar candidates in the US and Canada.

A report from the European Union Intellectual Property Office shows that the EU loses approximately €10.2 billion a year due to counterfeit medicines.

The time and resources required to finalize post-approval changes may be preventing manufacturers from modernizing facilities, or even scouting for new technology.

The company announced that it will build a new manufacturing facility at the GMR Aerospace Park at the Rajiv Gandhi International Airport in India.

Constantia Flexibles acquired a flexible packaging business from Lamp San Prospero SPA.

Xellia added laboratory space and personnel in Zagreb, Croatia to work on anti-infective products that combat the antimicrobial resistance problem.

A PESU membrane is now available for Sartorius Stedim Biotech Sartocon benchtop and production-scale filtration assemblies.

Allergan entered into a licensing agreement with AstraZeneca for MEDI2070, an anti-IL-23 monoclonal antibody in phase IIB development for the treatment of patients with moderate-to-severe Crohn’s disease.

The hospital received a five-year $5 million grant from CDC to survey for communicable diseases in children and evaluate vaccine effectiveness.

ViraTherapeutics will develop its VSV-GP therapy alone and in combination with other therapies.

PaizaBio will add aseptic injectable production capacity in Hangzhou, China.

Susan Schniepp, distinguished fellow at Regulatory Compliance Associates, discusses the assessment of risk in the processing of intravenous injectable drugs.

Laser-induced fluorescence, a rapid microbiology method for real-time airborne particle and microbial monitoring, enhances sterility assurance in pharmaceutical manufacturing.

As the pressure to bring drugs to market more quickly increases, companies are faced with the challenge of selecting the most effective cold-chain storage solutions.

High-purity low-endotoxin sugars improve robustness and stability of protein formulation and improve drug product quality.

Multiparticulates are increasingly used due to their flexibility in providing controlled-release, fixed-dose combinations, ease of taste-masking, and suitability for pediatric applications

Challenging molecules and markets are driving the development of new solutions for drug delivery.
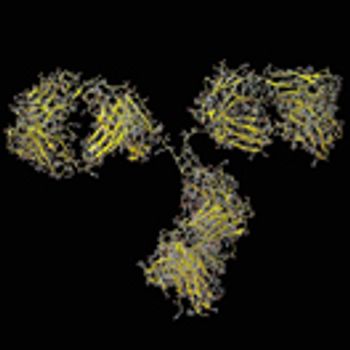
An approach to stabilize PBS-based formulations could provide a simple physiological solution for use of proteins in research, preclinical, diagnostics, and clinical studies, as well as commercial biotherapeutic products.

A new virus-retentive membrane may be used to filter chemical-defined cell culture media for risk mitigation.

Ravi Limaye gives an overview of the biosimilar industry and projects for 2020.

Onset expanded its product line for pharmaceutical cold-chain management with the launch two new products.

Wells Pharmacy Network is voluntarily recalling all of its products due to sterility concerns.

Researchers from the Wyss Institute explain a potential method for transporting and producing temperature-sensitive pharmaceuticals at a reduced cost.

R-Pharm Group, a private Russian pharmaceutical company, has opened a biopharmaceutical production plant in Yaroslavl, Russia, to produce biologics and biosimilars to treat autoimmune diseases and cancer. The facility, which has a line capacity is 2,000 L of cell culture per production cycle, is ready to be validated to US and Russia FDA and GMP requirements, the company reports.

Genentech’s biologics drug substance plant is the overall winner of the International Society for Pharmaceutical Engineering’s 2016 FOYA Awards.