
Under the terms of the partnership, Colorcon will work to accelerate i2O’s research and development efforts.

Under the terms of the partnership, Colorcon will work to accelerate i2O’s research and development efforts.

The investment will expand and upgrade manufacturing capacity and technology for pre-fillable syringes and will also includes a new manufacturing facility in Europe.
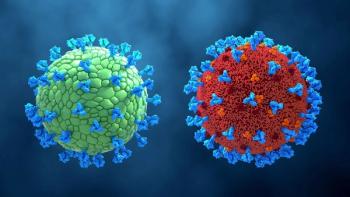
Analytical and functional characterization of virus-like particles enables process reproducibility and product consistency.

Scaling needs for potential COVID-19 vaccines depend not only on capacity, but also on supply chain challenges and technological hurdles.

After a difficult year, biopharma science delivers promising results.

The expansion will include new suites for the development and clinical manufacturing of drug product intermediates and drug products and cGMP suites for early-phase cGMP manufacture.

The companies have entered into a global collaboration and license agreement to develop zuranolone (SAGE-217) for major depressive disorder, postpartum depression (PPD), and SAGE-324 for essential tremor and other neurological disorders.

Wacker will support production of CureVac’s COVID-19 mRNA-based vaccine candidate at its biotech site in Amsterdam, with production scheduled to start in the first half of 2021.

Researchers concluded that the nasal spray should catch and coat the virus inside the nose, preventing it from spreading throughout the body, and it is then removed by nose-blowing or swallowing.

At the 2020 Bio/Pharma Virtual Congress, experts discuss the importance of characterizing excipients to ensure quality, safety, and effective performance.

The agreement will provide capacity for the manufacturing of AZD7442, currently being developed for the potential prevention and treatment of COVID-19, at Lonza's Portsmouth, NH site.

FDA published guidance for submitting standardized study data in electronic format.

Samsung Biologics has adopted Solentim’s cell seeding and cell metric platforms at its new R&D center in San Francisco, CA.

Orgenesis has completed its previously announced acquisition of Koligo Therapeutics with the additional acquisition of Tissue Genesis’ cell isolation technology, Icellator.

Bio/Pharma companies are galvanized to develop COVID-19 therapies under FDA’s acceleration program.

AFI representatives of the process validation working group explore and define key elements for an enhanced approach to process validation for sterile liquid and freeze-dried forms.

The IQ Consortium marks a decade of impactful innovation of collaboration through a network of pharmaceutical companies driving change for the bio/pharmaceutical industry.

Advances in preclinical development play a crucial role in reducing cost for developing biosimilars.

A data-integrity risk assessment tool has been developed for use with standalone R&D data-acquisition and processing software.

The acquisition will support the accelerated commercialization of Kyslecel (autologous pancreatic islets), Koligo’s personalized cell therapy for treating pancreatitis.

Moderna and Chiesi Group are collaborating on the discovery and development of mRNA therapeutics to treat pulmonary arterial hypertension.

The collaboration will expand the manufacturing capacity for lenzilumab, a COVID-19 therapeutic candidate, in advance of a potential Emergency Use Authorization in 2020.

The agreement will Increase global manufacturing capacity for Novavax’s vaccine candidate, NVX-CoV2373, to more than two-billion annualized doses when at full capacity in 2021.

The agreement between the two companies will significantly increase global supply capacity for Lilly's potential COVID-19 treatments.

The companies will focus on the development and commercialization of Seattle Genetics’ ladiratuzumab vedotin and TUKYSA (tucatinib).