
A case study investigated the root cause of failures in sterile filtration by evaluating the effects and interactions of four operating parameters.

A case study investigated the root cause of failures in sterile filtration by evaluating the effects and interactions of four operating parameters.

Multivariate data analysis can help biotech manufacturers deepen their process understanding.


Process monitoring ensures that the process performs within the defined acceptable variability that served as the basis for the filed design space.

The number of biotechnology-based human therapeutic products in the late-stage pipeline, and the average cost to commercialize a biotech product, have steadily increased. This has required biotech companies to use economic analysis as a tool during process development and for making decisions about process design. Process development efforts now aim to create processes that are economical, as well as optimal and robust.
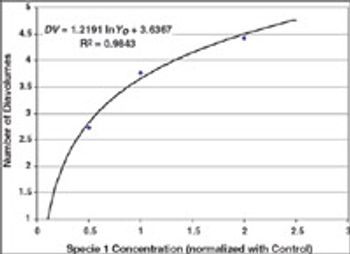
Case studies were run to test Process Analytical Technology applications for protein refolding, diafiltration, and cation exchange chromatography. It is shown that it is feasible to design control schemes that rely on measurement of product quality attributes and thereby enable real-time decisions.

Misinterpreting the effluent profiles obtained during tracer measurements performed for determining packing quality can often lead to excessively large percolation velocities and exaggeration of packing problems. Highly useful and reliable information can be obtained through characterization of tracer effluent curves using the method of moments, information that could be critical for successful scale-up of chromatographic steps. This is the sixth in the "Elements of Biopharmaceutical Production" series.

Creation and qualification of scale-down models is essential for performing several critical activities that support process validation and commercial manufacturing. This combined article is the fifth in the "Elements of Biopharmaceutical Production" series. Part 1 (March 2005) covered fermentation. In this segment, we present some guidelines and examples for scale-down of common downstream unit operations used in biotech processes - chromatography and filtration.

Lot-to-lot variations between raw materials can greatly impact process performance.
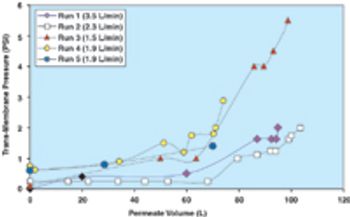
Filtration is one of the most commonly used unit operations in the manufacturing of biopharmaceuticals. This is the second part of the fourth article in the "Elements of Biopharmaceutical Production" series. In this second segment, Manoj Menon and Frank Riske present an approach for the development and optimization of a TFF application, followed by a contribution from Jennifer Campbell and Elizabeth Goodrich reviewing key issues involved in validation of a TFF step.
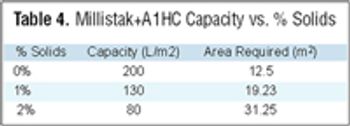
Filtration is one of the most commonly used unit operations in biopharmaceutical manufacturing. Available formats include direct or normal flow filtration (NFF) and cross or tangential flow filtration (TFF). These methods are used for sterilization and virus filtration, depth filtration or ultrafiltration, and diafiltration applications. Some common objectives include:

Operational planning and early cost analyses are key to generating optimal, robust, and economical commercial processes.