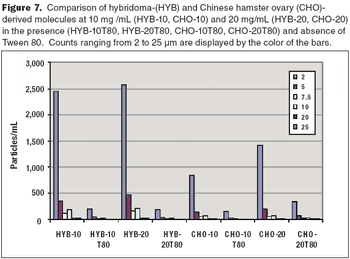
How to maintain product stability and prevent particulates.

How to maintain product stability and prevent particulates.
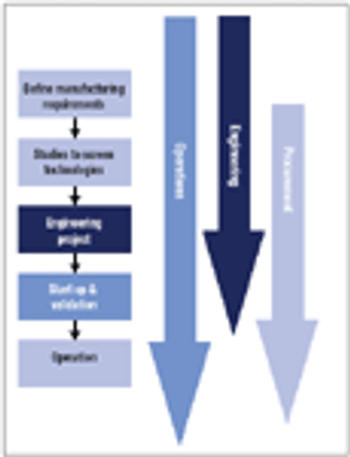
In new disposables projects, it is critical that engineering, procurement, and operations groups work together early on to manage supply chain risk.
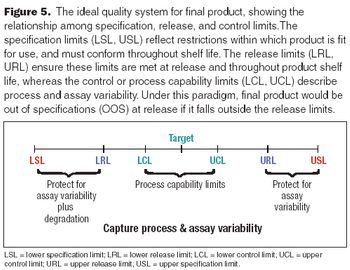
Set limits to provide incentives for process improvements.
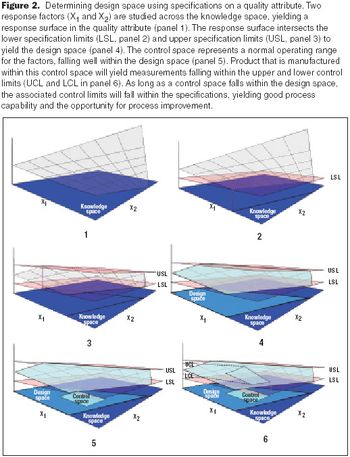
Resolve confusion about measurements.