
The complexity of newer biotherapeutic molecules calls for alternative drug delivery strategies.

The complexity of newer biotherapeutic molecules calls for alternative drug delivery strategies.

Speed-to-market, capacity, technology development, and increased investment top the list.
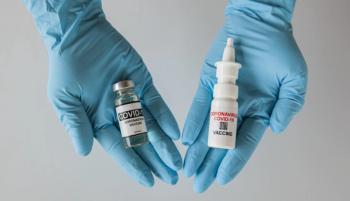
The intranasal route of administration is showing clinical promise, particularly for COVID-19, but there are multiple hurdles to overcome to ensure successful formulation.

Large-scale single-use bioreactors prove successful as limits are tested.

SUB bioreactor performance has had seen its limits and successes.

Early control architecture decisions can impact a facility’s long-term growth, flexibility, and efficiency.

Establishing a data management strategy in-lab is a first step in generating successful analytical studies.

Public health challenges have highlighted the need for agility in maintaining the quality of medicines.
Establishing bioassay studies for biosimilar development is important for supporting regulatory filings.

A new program will test the safety and suitability of new inactive ingredients to encourage the accelerated adoption of FDA-accepted excipients in drug development.

Change of roster does not change the goal line. We are all patients first.

Susan J. Schniepp, distinguished fellow at Regulatory Compliance Associates, answers some commonly asked questions about aseptic processing.