
Emergence of advanced manufacturing technology to ensure quality of biopharmaceutical drugs combined with efforts to identify a regulatory pathway indicate that a distributed manufacturing model is within reach.

Emergence of advanced manufacturing technology to ensure quality of biopharmaceutical drugs combined with efforts to identify a regulatory pathway indicate that a distributed manufacturing model is within reach.
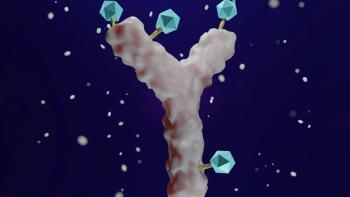
There is no such thing as a perfect linker.

Re-evaluating the basics of mAb production may be beneficial for the biopharma industry as a whole.

USP offers strategies to minimize residual impurities in downstream processing.

The implementation of Industry 4.0 is projected to save time and cost for designing an optimal manufacturing process.
Interest grows in AI, digitalization, robotics, and sustainability.

A look at using best practice tech transfer methods for CGTs to increase process and analytics robustness while being scalable.

Industry investments indicate increasing trust in the newest modalities, including mRNA approaches.

There are key elements that should be covered when dealing with third-party auditors, according to Siegfried Schmitt, vice president, Technical, at Parexel.