
Susan Schniepp, distinguished fellow at Regulatory Compliance Associates, discusses the assessment of risk in the processing of intravenous injectable drugs.

Susan Schniepp, distinguished fellow at Regulatory Compliance Associates, discusses the assessment of risk in the processing of intravenous injectable drugs.

Laser-induced fluorescence, a rapid microbiology method for real-time airborne particle and microbial monitoring, enhances sterility assurance in pharmaceutical manufacturing.

As the pressure to bring drugs to market more quickly increases, companies are faced with the challenge of selecting the most effective cold-chain storage solutions.

High-purity low-endotoxin sugars improve robustness and stability of protein formulation and improve drug product quality.

BioPharm International editors seek bioprocessing experts to contribute technical articles in 2017.

Multiparticulates are increasingly used due to their flexibility in providing controlled-release, fixed-dose combinations, ease of taste-masking, and suitability for pediatric applications

Challenging molecules and markets are driving the development of new solutions for drug delivery.
An approach to stabilize PBS-based formulations could provide a simple physiological solution for use of proteins in research, preclinical, diagnostics, and clinical studies, as well as commercial biotherapeutic products.

Efforts to accelerate drug development will alter fee structure and require ready production sites.
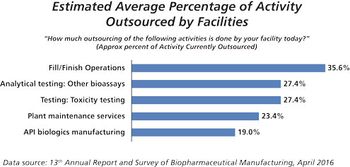
This key bioprocessing segment is expecting continued growth.

A new virus-retentive membrane may be used to filter chemical-defined cell culture media for risk mitigation.
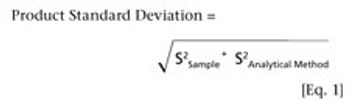
Knowing how method performance impacts out-of-specification rates may improve quality risk management and product knowledge.

The Print & Check Flex Machine from Antares Vision is a flexible high-capacity serialization unit created for track and trace.

The CELLdisc from Greiner Bio-One is a multilayer cell culture vessel that offers a growth area of up to one square meter for adherent mammalian cells.

Click the title above to open the BioPharm International October 2016 issue in an interactive PDF format.