
This article reviews systems and processes that enable a laboratory to approach troubleshooting in an effective way, while also taking a proactive, preventive approach to managing atypical laboratory scenarios.

This article reviews systems and processes that enable a laboratory to approach troubleshooting in an effective way, while also taking a proactive, preventive approach to managing atypical laboratory scenarios.
The success of a truly integrated continuous processing platform relies on the collaborative efforts of upstream and downstream specialists.
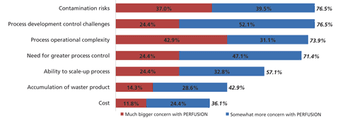
Although widespread adoption of continuous bioprocessing has been slow, some processes have been an exception.
This article summarizes the approaches, challenges, and future perspectives for the characterization of N-glycans in biopharmaceutical products.

Approval of breakthrough therapies requires expedited quality assessment.

Informing your clients of possible changes in equipment is imperative when upgrading a laboratory, says Susan Schniepp, distinguished fellow at Regulatory Compliance Associates.

As more companies decouple drug substance from finished drug manufacturing operations, an integrated approach can ensure safe, reliable logistics for frozen storage and shipping.

New reports indicate that drug prices are slowing compared to other healthcare costs.

Increased understanding of potential impurities has spurred efforts to standardize monitoring procedures.

How statistical methods and novel indices can be used to monitor and benchmark variability, to guide continuous improvement programs.
An increase in biologics raises awareness of particle generation and its role in negative patient outcomes.

Click the title above to open the BioPharm International June 2017 issue in an interactive PDF format.