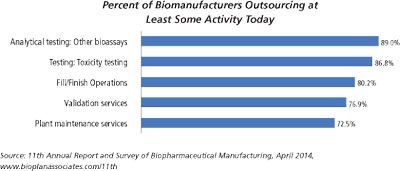
A tremendous amount of analytical testing is required to support a biopharmaceutical product from discovery, development, and clinical trials, through manufacturing and marketing. Numerous methods are used to fully characterize large molecules because of their complexity-characterizing them is significantly more difficult than it is for small molecules. Biopharmaceuticals are produced via living systems, i.e., E. coli, yeast, or mammalian cells, which require additional testing matrices.