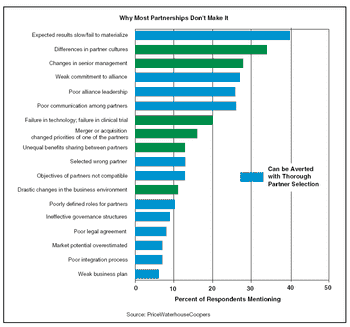
Successful acquisition of funding from venture capital (VC) partnerships requires a carefully planned, well-executed marketing campaign. You are, after all, selling ownership interests in your company when you seek VC investors. This sale requires an attractive product, nicely packaged, and presented in a way that allows the potential customer to understand and appreciate its value. The attractiveness of your company is the sum of many factors including revenue potential, intellectual property estate, time-to-milestones, existing competition, ease of entry, quality of management, and the perceived interest in your work by "the big guys" in your sector. Every potential venture investor will not perceive your company the same way.