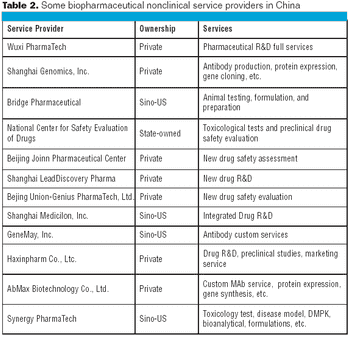
Sir Isaac Newton said, back in 1676, "If I have seen further, it is by standing on the shoulders of Giants." Newton's modest self-assessment of his scientific competence holds relevance in today's debate on the value of scientific meetings, and our quest for faster scientific discovery.