
Knowing and addressing regulatory expectations early on can avoid unexpected delays later, says Siegfried Schmitt, principal consultant at PAREXEL.

Knowing and addressing regulatory expectations early on can avoid unexpected delays later, says Siegfried Schmitt, principal consultant at PAREXEL.

This article highlights 15 years of changes in biopharmaceutical manufacturing.

After 30 years of biologic-drug advances, the industry and patients still have a lot to learn.

Biopharma companies can balance competing demands from patients, investors, and regulators by keeping a focus on science.

Modern technologies, including Industry 4.0 and the Industrial Internet of Things, offer opportunities to increase biopharmaceutical manufacturing efficiency.

FDA seeks more efficient testing to spur development of less costly biotech therapies.

Process analytical technology tools have enabled manufacturers to monitor and control their production processes.

Quality experts share insights on what the future may hold regarding regulatory quality requirements for biopharmaceuticals.

BioPharm International asked an FDA spokesperson how the agency plans on handling these issues in the future.

Increasing demand for biologics is driving the need for innovation in bioprocessing.
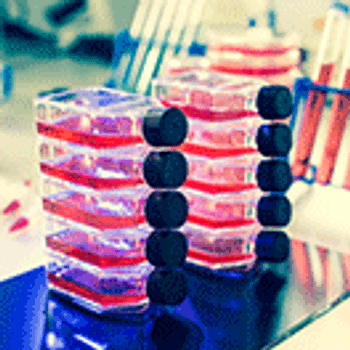
Late-stage and commercial biomanufacturing pose a challenge to cell-culture processing.

Biosimilars and biobetters face developmental challenges to achieving commercialization.

Safeguarding the know-how behind biopharmaceutical innovation is crucial to the industry’s future, but, in the US, some argue it is becoming increasingly difficult to do.

Click the title above to open the BioPharm International July 2018 issue in an interactive PDF format.