
FDA draft guidances seek to maintain accurate drug information in new media.

FDA draft guidances seek to maintain accurate drug information in new media.

As payers refuse to cover new drugs, CMOs take a hit.

Microbioreactors Increase Productivity

Quantitative Mycoplasma DNA Offers Quality Control
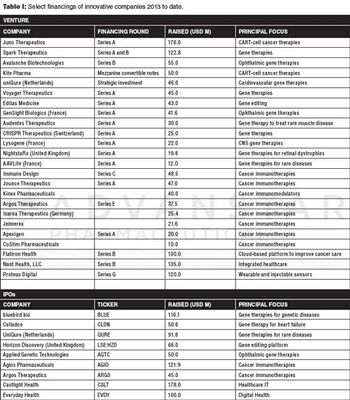
Gene therapy, immune-oncology, and digital healthcare technologies offer investors promise for innovation investments.
Establishing the CQAs of a mAb product by evaluating impact and uncertainty during risk assessment.
Light scattering analysis combined with more rapid size exclusion chromatography improves protein characterization.

Is there potential for growth in Brazil's phytotherapic drug market?

Stakeholders face challenges and benefits from a more secure pharmaceutical supply chain.

Supplier Audit Program Marks Progress

Achieving excellence in human performance is an achievable system of cultivating and sustaining exceptional levels of performance.

With globalization, biopharmaceutical companies must establish strategies to minimize vulnerabilities in the raw-materials supply chain.

Challenges in Securing the Biopharma Supply Chain

Biochemistry Analyzers Improve Accuracy

Click the title above to open the BioPharm International July 2014 issue in an interactive PDF format.