
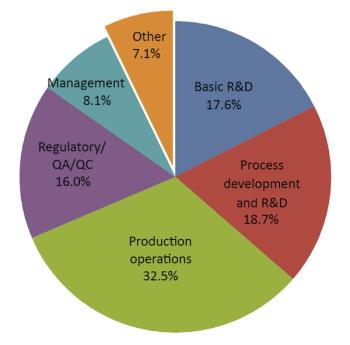
The results of BioPharm International's fourth annual salary survey, conducted in the fall of 2009, shows that women lag behind their male counterparts.

The results of BioPharm International's fourth annual salary survey, conducted in the fall of 2009, shows that women lag behind their male counterparts.

BiondVax Pharmaceuticals Ltd, an Israeli biopharmaceutical company, has completed Phase 1-2 clinical trials of its Multimeric-001 universal flu vaccine.

Sanofi Pasteur, the vaccines division of the Sanofi-Aventis Group, has signed an agreement with KaloBios Pharmaceuticals for the development of humanized antibody fragment to both treat and prevent Pseudomonas aeruginosa infections.

Genzyme (Cambridge, MA) will outsource fill-and-finish operations for its drugs Cerezyme, Fabrazyme, Myozyme, and Thyrogen to Illinois-based Hospira, Inc.
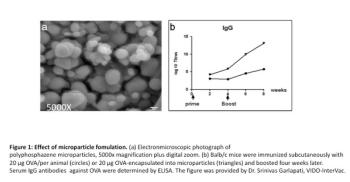
This article presents ongoing research at VIDO-InterVac for the development of safer and more effective adjuvants. These include adjuvants that stimulate innate immunity in the body and combination adjuvants.

MedImmune, a division of AstraZeneca (London, UK), yesterday recalled 13 lots (4.7 million doses) of its nasal spray H1N1 (swine) flu vaccine, because of a drop in potency.


Merck & Co. Inc., (Whitehouse Station, NJ) has announced plans to buy the biologics business of the Avecia group (Tees Valley, UK), through a Merck affiliate (Merck Sharp & Dohme, MSD).

Sanofi Pasteur, the vaccines division of France-based Sanofi-Aventis Group, is voluntarily recalling about 800,000 doses of H1N1 (swine) flu vaccine meant for children between the ages of 6 months and 35 months because quality assurance tests indicate the vaccine doses lost some potency.

GlaxoSmithKline Biologicals SA (GSK, London, UK) and Intercell (Vienna, Austria) have formed a strategic alliance to accelerate the development and commercialization of needle-free, patch-based vaccines.

Artes Biotechnology (Langenfeld, Germany) and Rhein Minapharm Biogenetics (Cairo, Egypt) have produced a new target of a therapeutic protein.

Novo Nordisk has initiated its first Phase 1 clinical trial with an oral insulin analog, NN1952, which was created using Merrion Pharmaceuticals' GIPET technology.

When the CMC Biotech Working Group began developing a case study on applying Quality by Design (QbD) to biotech products, the goal was to challenge conventional thinking on the subject.

Gyros AB (Uppsala, Sweden), a provider of automated micro-immunoassays for therapeutic protein development, has signed a distribution agreement with DI Biotech (Seoul, Korea), a supplier to the Korean life sciences market.

A ground breaking ceremony for the commencement of construction of the National Institute for Bioprocessing Research and Training (NIBRT) at Belfield Innovation Park in Dublin was held on November 29.

Sinovac Biotech Ltd. (Beijing, China), a developer and provider of vaccines, has executed a joint venture agreement to establish Sinovac (Dalian) Vaccine Technology Co., Ltd. (Sinovac Dalian).

Genzyme Corporation (Cambridge, MA) has begun shipping vials of newly produced Cerezyme (imiglucerase for injection) manufactured at its plant in Allston Landing, MA.

The German fill-finish provider Vetter inaugurated a new US facility in Skokie, IL, just outside Chicago, along with a new subsidiary, Vetter Development Services USA Inc.

The US Food and Drug Administration has approved Agriflu, a new seasonal flu vaccine manufactured by Novartis Vaccines and Diagnostics (Siena, Italy), for people ages 18 years and older to prevent disease caused by influenza virus subtypes A and B.

The Swiss pharmaceutical company Novartis has opened a new vaccine manufacturing facility in Holly Springs, NC.

Following a five-week inspection of Genzyme's plant in Allston Landing, MA, the US Food and Drug Administration is still not satisfied with the company?s large-scale manufacturing of Lumizyme.

The FDA and Genzyme have notified healthcare professionals about the potential for foreign particle contamination of several Genzyme products: Cerezyme, Fabrazyme, Myozyme, and Thyrogen, which are supplied as lyophilized powders requiring reconstitution before administration; and Aldurazyme, which is supplied as a liquid solution. The foreign particles include stainless steel fragments, non-latex rubber from the vial stopper, and fiber-like material from the manufacturing process, and could potentially cause serious adverse events in patients.

Abraxis Biosciences, a Los Angeles-based biotechnology company, unveiled a $70-million biopharmaceutical manufacturing plant in Phoenix, to serve as the company's manufacturing site for its pipeline of biologics.

FDA Publishes Guidance on Diagnostic Tests for 2009 H1N1 Influenza Virus

Morphotek, Inc. (Exton, PA), a subsidiary of Eisai, Inc., has entered into a license agreement with Centocor Ortho Biotech, Inc. (Horsham, PA), for the development and commercialization of an antibody that targets a tumor-associated antigen over-expressed in many solid tumors.

SAIC-Frederick, Inc. (Frederick, MD), has signed a contract with GenVec, Inc. (Gaithersburg, MD), for the development of influenza and HIV vaccines in support of the Vaccine Research Center (VRC, Frederick, MD).

Octapharma Group (Bad Homburg, Germany) and Fresenius Kabi (Lachen, Switzerland) have signed a license, development, and supply agreement for the use of Fresenius Kabi's HESylation technology to develop a HESylated recombinant protein.

The US Food and Drug Administration has granted priority review for Shire plc's (Cambridge, MA) New Drug Application (NDA) for velaglucerase alfa, the company's enzyme replacement therapy in development for the treatment of Type 1 Gaucher disease.

Brain drug delivery company to-BBB, (Leiden, The Netherlands) has entered into a research collaboration with Genzyme Corporation (Cambridge, MA) to evaluate to-BBB?s proprietary G-Technology to enhance delivery of biologics for neurodegenerative diseases.