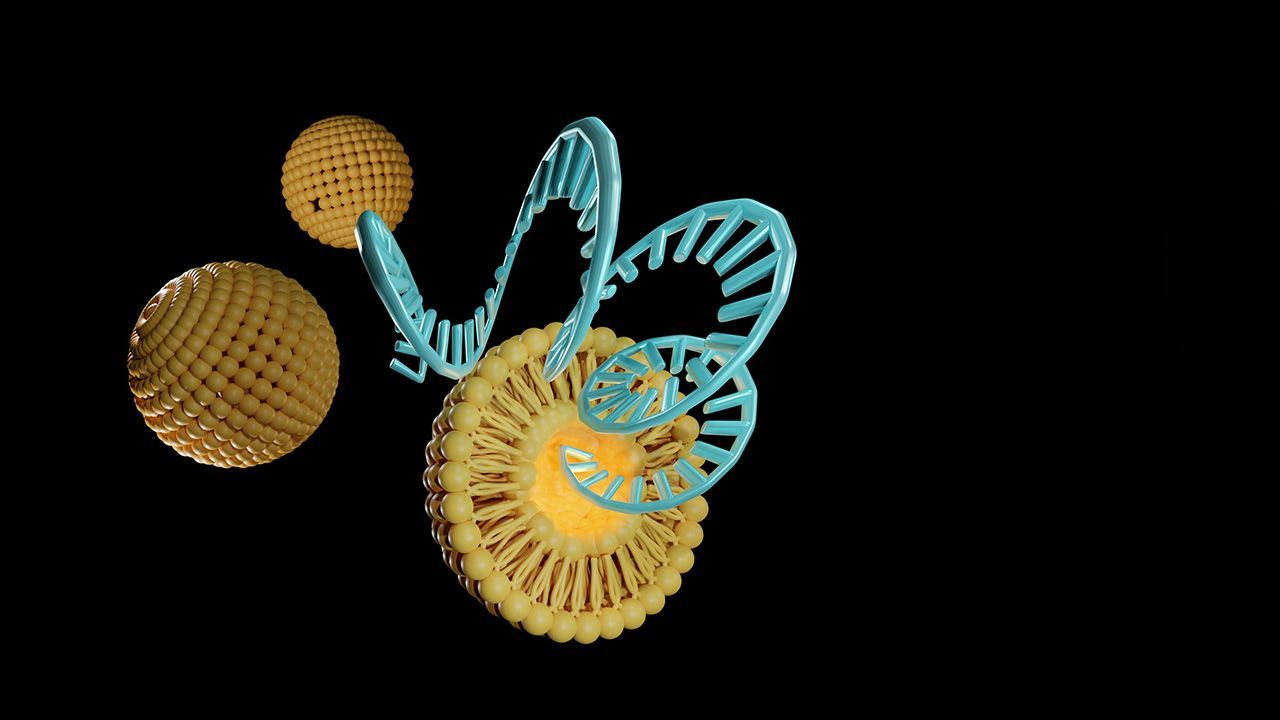
Vaccine Development and Manufacturing
Latest News
Latest Videos

Shorts

More News

Regulators and manufacturers face new uncertainty as shifting CDC vaccine language raises concerns for evidence-based oversight and biopharma compliance.

Cellevate’s nanofiber technology enables higher viral vaccine titers in adherent cell cultures, advancing efficiency in large-scale biomanufacturing.

Naobios and Olon France create custom HCP ELISA to advance vaccine trials and strengthen global biopharmaceutical manufacturing capacity.
ACIP votes to separate MMR and varicella vaccines (MMRV) for children to cut febrile seizure rates.
Moderna’s updated Spikevax and mNEXSPIKE COVID-19 vaccines target the LP.8.1 variant of SARS-CoV-2 for high-risk groups and older adults.

FDA will review GSK’s application to expand the use of its respiratory syncytial virus vaccine, Arexvy, to adults aged 18–49 who are at increased risk.

The next-generation monoclonal antibody could potentially be used to prevent and treat active COVID-19 infections, according to its developer, providing a non-vaccine option.

The spray-dried formulations for Ethris’ mRNA vaccine candidates will be developed at Lonza’s Bend, Ore., Center of Excellence in accordance with GMP standards.

The partnership will advance the accessibility, scalability, and affordability of RNA-based vaccines.

Pointing to the continued circulation and evolution of COVID-19, WHO published a statement on the COVID-19 vaccine antigen composition to respond to variants of the virus.

Using Orexo’s powder-based drug delivery technology, the companies will develop mucosal vaccines in an inhaled formulation.

This article looks at how the MAPPs assay works, how it integrates with other immunogenicity assays, and just how it can be flipped to help in vaccine design.

Pfizer’s bivalent RSV prefusion F vaccine has been approved by FDA to prevent lower respiratory tract disease resulting from the respiratory syncytial virus in adults 18 to 59 years of age who are at increased risk.

Evonik's plant-based squalene, PhytoSquene, used in parenteral drug delivery applications, won the CPHI Excellence in Pharma Award in the “Sustainability” category.
There is potential to effectively marry the best aspects of mRNA as a modality, to other existing or nascent exciting platforms, such as gene editing.

The agency is recommending the extension of the smallpox and mpox vaccine, Imvanex, to adolescents aged 12 to 17.

A coordinated and international response is needed to help control the latest mpox outbreak in Africa.

The approvals and authorizations come amid a late summer surge in the virus that is tracking a bit earlier than the early fall wave in the US in 2023.

National Institutes of Health’s National Institute of Allergy and Infectious Diseases collaborated with the University of Sciences, Techniques and Technologies of Bamako, Mali to conduct the trials.

The virus, which is contagious and a common cause of respiratory illness, hospitalizes up to 160,000 people and is responsible for as many as 13,000 deaths in the United States every year.

The companies have restructured their existing collaboration into a licensing agreement that invests in mRNA development.

The companies have completed the expansion and qualification phase and now enter a stand-by phase for five years.

An ASGCT Panel titled Novel Nucleic Acid and Cell-based Vaccines for Cancer highlights promising mRNA personalized cancer vaccine work.
This research focuses on refining the clarification step by assessing the performance of various depth filters. The study explores both conventional cellulose-based and innovative synthetic depth filters, utilizing pneumococcal fermentation harvests from two challenging serotypes.

The outcomes of a workshop organized by WHO and ICMRA regarding COVID-19 vaccine strain updates are presented in a report published by regulators.