
The pharmaceutical industry grows despite conflict in the Middle East.

The pharmaceutical industry grows despite conflict in the Middle East.

Practicality of implementation should be a part of vision in the bio/pharmaceutical industry.
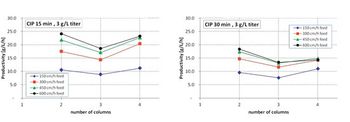
A twin-column periodic countercurrent capture process that combines an efficient sequential, countercurrent loading process with a minimal twin-column hardware configuration is described.

Through its educational and networking opportunities, the American Association of Pharmaceutical Scientists plays an important role in partnering throughout the drug- development and commercialization process.

Manufacturing standards are considered key to preventing drug recalls and shortages.

The authors review the angiopoietin pathway as an alternative for safer and more efficacious anti-angiogenic therapeutics.

Foreign companies zero in on Myanmar with the hope of securing a foothold in its pharmaceutical market.

By embracing efficiency and quality, biopharmaceutical organizations can work better and achieve better work.

Early-stage companies are now embraced by buyers of new issues.
A well-designed comparability study can demonstrate the performance and advantages that can be gained when adopting a new protocol.
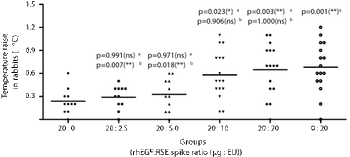
The correlation between limulus amebocyte lysate (LAL) assay and rabbit pyrogen test (RPT) targeting recombinant human epidermal growth factor (rhEGF) as active molecule was assessed.

Click the title above to open the BioPharm International October 2013 issue in an interactive PDF format.