
Sponsored Content
This white paper examines better solutions for flexible biopharmaceutical production sites and process designs that meet the needs that outdated traditional cleanroom facility and infrastructures can no longer provide.

Sponsored Content
This white paper examines better solutions for flexible biopharmaceutical production sites and process designs that meet the needs that outdated traditional cleanroom facility and infrastructures can no longer provide.
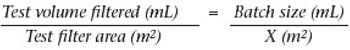
Why staining is crucial in flow decay studies.
Biopharmaceutical processes typically require a significant investment in equipment-often a substantial obstacle for start-up companies. The risk of drug development failure is often high, further limiting access to the required capital. Flexibility and lower capital outlays are required not only by start-up companies, but also by research organizations with multiple product lines and by companies requiring quick capacity increases. Disposable technologies offer the highest potential for these companies to meet their business requirements. With lower capital requirements and increased flexibility, disposables are an important part of these companies' risk management strategy.
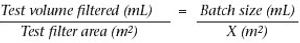
September 1st 2009
November 2nd 2006