The authors outline cell-line development and process scale-up for an antibody program in which the antibody requires additional processing by a site-specific enzyme for correct functionality.

The authors outline cell-line development and process scale-up for an antibody program in which the antibody requires additional processing by a site-specific enzyme for correct functionality.
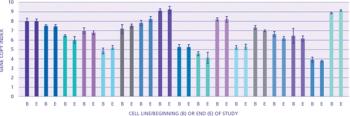
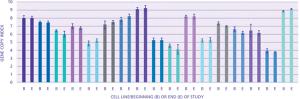
The author describes expression technology that produces cell lines with high genetic stability.
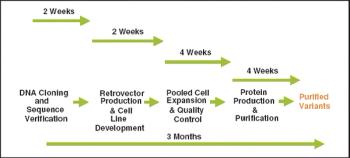
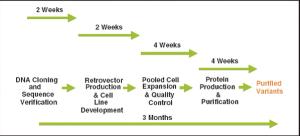
The speed at which a recombinant protein product progresses into clinical trials is of vital importance for both small biotechnology companies as well as the biopharma groups of large pharmaceutical companies. For mammalian cell lines, two major impacts on the project timeline are the ability to quickly identify a product candidate and subsequently produce a high-expressing cell line for that product. The advent of various computer-based protein design methodologies and antibody discovery technologies for developing protein therapeutics has resulted in large numbers of protein or antibody variants that must be screened to identify the best clinical candidate.
Published: May 1st 2012 | Updated:
Published: February 9th 2006 | Updated:
Published: September 1st 2016 | Updated: