|Articles|March 15, 2020
A Case Study in Pharmacopoeia Compliance: Excipients and Raw Materials
Author(s)J. Mark Wiggins, Joseph A. Albanese
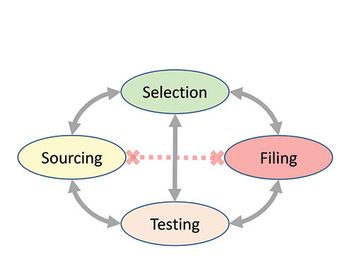
This case study is based on the experience of one of the authors but is applicable to all companies across the broader industry, illustrating the potentially surprising point that some compliance difficulties may be of the company’s own making.
Advertisement
Newsletter
Stay at the forefront of biopharmaceutical innovation—subscribe to BioPharm International for expert insights on drug development, manufacturing, compliance, and more.
Advertisement
Advertisement
Advertisement
Trending on BioPharm International
1
How CDMO Alliances Can Provide End-to-End Service that Reduces Drug Development Time and Costs
2
FDA Clears PharmaResearch IND for Nano-Based Cancer Drug PRD-101
3
First-in-Human Study Validates Safety of Next-Generation mRNA–LNP Platform
4
Industry Outlook 2026: Key Forces Transforming Drug Pipelines (Part 2)
5