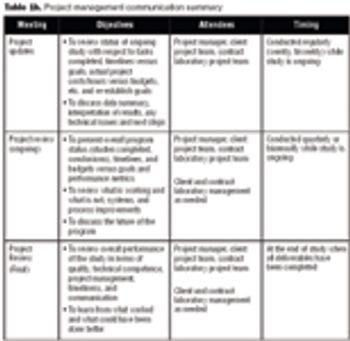
Are the deadlines for your outsourced projects often not met? Are you unsure of the status of your project at any given time? Is the original budget of your study typically exceeded?

Are the deadlines for your outsourced projects often not met? Are you unsure of the status of your project at any given time? Is the original budget of your study typically exceeded?

Partnering with a surging number of CROs, CMOs, CSOs, and other niche providers, biopharm companies in 2007 will have an estimated spend of more than $7 billion on international clinical trial outsourcing alone.

Consider the number of patients, the dose each will receive (and how this dose is calculated), the number of doses per patient, and what overage is needed to allow for vial breakage.
The greatest benefits of outsourcing are realized when a company takes a strategic approach rather than a tactical approach.

The contract manufacturer must have sufficient capacity so it can absorb possible surge in demand, and back-up capability in case of a power failure or other event.

A tremendous amount of analytical testing is required to support a biopharmaceutical product from discovery, development, and clinical trials, through manufacturing and marketing. Numerous methods are used to fully characterize large molecules because of their complexity-characterizing them is significantly more difficult than it is for small molecules. Biopharmaceuticals are produced via living systems, i.e., E. coli, yeast, or mammalian cells, which require additional testing matrices.