
Ben Edwards of Avance Clinical explains how generative AI and adaptive trials speed early-stage drug development by improving data access and capital efficiency.
Feliza Mirasol is the science editor for BioPharm International.

Ben Edwards of Avance Clinical explains how generative AI and adaptive trials speed early-stage drug development by improving data access and capital efficiency.

As the J.P. Morgan Healthcare Conference gets underway, Lonza’s Michael De Marco weighs in on regional manufacturing shifts as biotech pipelines mature, signaling new therapy launches within 12–18 months.

In part two of an interview, Daniel Delubac, iOrganBio CEO, explains how digitized, in-vitro human biology models aim to improve preclinical drug testing.

AI-driven human cell models aim to replace animal testing and deliver more accurate, data-driven insights, says iOrganBio CEO Daniel Delubac ahead of the J.P. Morgan Healthcare Conference.

EU approval of a prefilled Shingrix syringe streamlines vaccine delivery, while Phase III data position bepirovirsen as a potential functional cure for hepatitis B.
In a new team-up, MD Anderson and SOPHiA GENETICS will apply advanced analytics to cancer testing to improve complex data interpretation for clinical use.

Digitalization is expected to reshape bioprocessing, supply chains, and quality systems for biopharma developers amid tariffs, geopolitics, and shifting capital flows, according to BioPlan Associates’ Eric Langer.

The CDC has narrowed pediatric vaccine guidance, resetting evidence expectations and signaling potential shifts in vaccine uptake and market demand.
China’s authorization of GSK’s Nucala (mepolizumab) for treating COPD signals regulatory momentum for precision respiratory medicines in the country.
The company’s Phase III setrusumab trials in osteogenesis imperfecta missed fracture endpoints but showed strong bone density gains.

Key 2025 FDA draft and final guidances emphasize the modernization of biotech regulations, acceleration of rare-disease therapies, and streamlining of biosimilar pathways.

Autolomous and Trenchant’s partnership, begun early in 2025, addresses unmet CGT manufacturing needs, including slashing costs and cutting timelines.

A look at interchangeable biosimilars gaining FDA approval, the growth in global partnerships, and the rising biologics patent cliff.

Samsung Biologics’ Maryland acquisition signals a major push into US biologics manufacturing, reshaping the company’s capacity strategy and supply chain resilience.

Advances in precision analytics have pushed the needle forward in characterizing and assessing the safety and quality of next-gen biologics.
This past year in biologics was marked by clinical trends, manufacturing scalability, and regulatory acceleration for antibody therapeutics.

Norgine has gained regional rights to Vir Biotechnology’s hepatitis delta drug pairing, strengthening late-stage antiviral development across Europe, Australia, and New Zealand.
European regulators authorize AstraZeneca’s anifrolumab pen for lupus, reinforcing industry momentum toward self-injected biologics and decentralized immunology care.

Fondazione and Orphan Therapeutics outline a US access model for Waskyra, highlighting new approaches to manufacturing, distribution, and sustainability.

Charles River has launched a second cohort to speed CGT innovation with technical guidance and scalable manufacturing support.
The new GSK–Oxford BioTherapeutics partnership capitalizes on proteomics-driven oncology target discovery for advancing antibody therapeutics development.

FDA’s approval of the first Wiskott-Aldrich gene therapy signals a shift in manufacturing and regulatory standards for rare-disease CGTs.

FDA’s clearance of a chemically enhanced stem cell therapy for severe aplastic anemia drives forward cell therapy development and manufacturing innovation.

Priority review, RMAT, and breakthrough status were key regulatory actions intensely accelerating advanced next-gen therapy development in 2025.
FDA’s new-indication approval for lisocabtagene maraleucel in marginal zone lymphoma boosts engineered T-cell treatment for relapsed lymphoma.
Under the expanded agreement, the companies are targeting amyloid removal strategies to address unmet needs in rare diseases.
siRNA-based cell reprogramming shows potential to improve metabolic health with durable effects, signaling a notable development for future therapeutic strategies.
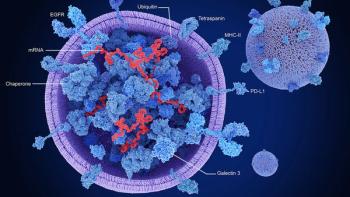
Engineered exosomes could reshape therapeutic development by redefining delivery, manufacturing models, and regulatory standards.

FDA outlines reduced primate testing for monoclonal antibodies, signaling a shift toward modern evaluation tools that may influence drug development practices.